ISSN: 1839-9940
J Genomics 2024; 12:19-25. doi:10.7150/jgen.90833 This volume Cite
Research Paper
Molecular identification and whole genome sequence analyses of methicillin-resistant and mastitis-associated Staphylococcus aureus sequence types 6 and 2454 isolated from dairy cows
1. Department of Microbiology and Hygiene, Bangladesh Agricultural University, Mymensingh-2202, Bangladesh.
2. Veterinary Medicine and Food Security Research Group, Medical Laboratory Sciences Program, Faculty of Health Sciences, Abu Dhabi Women's Campus, Higher Colleges of Technology, Abu Dhabi, UAE.
3. Institute of Hygiene and Environmental Medicine, Justus Liebig University Giessen, Biomedical research center Seltersberg, Schubertstrasse 81, 35392 Giessen, Germany.
4. National Institute of Biotechnology, Savar, Dhaka, Bangladesh.
5. Department of Integrative Biology, College of Arts and Sciences, University of South Florida, St. Petersburg, Florida, USA.
# Mohammad H. Rahman and Mohamed E. El Zowalaty are equal first authors and were arranged by order of increasing seniority.
Received 2023-10-7; Accepted 2023-12-19; Published 2024-1-18
Abstract
The emergence of antimicrobial-resistant and mastitis-associated Staphylococcus aureus is of great concern due to the huge economic losses worldwide. Here, we report draft genome sequences of two Staphylococcus aureus strains which were isolated from raw milk samples obtained from mastitis-infected cows in Bangladesh. The strains were isolated and identified using conventional microbiological and molecular polymerase chain reaction (PCR) methods. Antibiotic susceptibility testing was performed. Genomic DNA of the two strains was extracted and the strains were sequenced using the Illumina NextSeq 550 platform. The assembled contigs were analyzed for virulence determinants, antimicrobial resistance genes, extra-chromosomal plasmids, and multi-locus sequence type (MLST). The genomes of the two strains were compared with other publicly available genome sequences of Staphylococcus aureus strains. The raw read sequences were downloaded and all sequence files were analyzed identically to generate core genome phylogenetic trees. The genome of BR-MHR281strain did not harbour any antibiotic resistance determinants, however BR-MHR220 strain harbored mecA and blaZ genes. Analysis of BR-MHR220 strain revealed that it was assigned to sequence type (ST-6), clonal complex (CC) 5 and spa type t304, while BR-MHR281 strain belonged to ST-2454, CC8, and harbored the spa type t7867. The findings of the present study and the genome sequences of BR-MHR220 and BR-MHR281 strains will provide data on the detection and genomic analysis and characterization of mastitis-associated Staphylococcus aureus in Bangladesh. In addition, the findings of the present study will serve as reference genomes for future molecular epidemiological studies and will provide significant data which help understand the prevalence, pathogenesis and antimicrobial resistance of mastitis-associated Staphylococcus aureus.
Introduction
Bovine mastitis is a multi-factorial, multi-etiological, highly contagious common livestock production-related disease. Bovine mastitis causes huge economic losses and leads to great implications in dairy industry worldwide due to the reduced milk quality and quantity in dairy herds [1]. Mastitis is a very complex and multi-etiological disease caused by more than 140 species of bacteria [2].
Staphylococcus aureus (S. aureus) is one of the major foodborne pathogens associated with various human infections and animal diseases including important livestock such as cattle, cows, sheep and goats [3]. S. aureus is the most common etiological agent associated with bovine mastitis worldwide and results in a range of manifestations, including a large proportion of subclinical and chronic infections [4,5]. Among livestock, cows are a common reservoir of S. aureus, and dairy cattle frequently experience clinical and subclinical mastitis due to S. aureus intramammary infections [6].
S. aureus possesses an arsenal of virulence and antimicrobial resistance determinants which are subject to horizontal genetic transfer and recombination [7]. Genome sequencing has provided insight into the genotypic features of various S. aureus clones worldwide, delivering more options for developing therapeutics and molecular diagnostic tools to detect resistant and difficult-to-treat strains.
Genome sequencing and characterization of S. aureus isolated from bovine milk is an important tool in the epidemiological studies of bovine mastitis. They can provide clinically relevant results and contribute to the understanding of the pathogen's dissemination and contagious properties [4,8].
The detection of antimicrobial resistant S. aureus strains isolated from bovine mastitis, its zoonotic potential, and the possibility of transmission to humans via the consumption of raw unpasteurized dairy and livestock products are increasing public health concerns [9,10]. Irrational use of antibiotics in bovine mastitis treatment may results in the development of resistant strains and residual antibiotics in milk also pose serious public health concerns [9,11]. The factors, etiologies, treatment, and molecular characterization of common bovine mastitis-causing pathogens were recently reported [12], yet no studies on the application of whole genome sequencing in mastitis-causing pathogens were reported from Bangladesh. Here we report the draft genome sequences of two S. aureus strains isolated from raw milk samples obtained from mastitis lactating cows in Bangladesh.
Materials and Methods
Ethics statement
The study protocol entitled "Development of polyvalent mastitis vaccine and probiotics for prevention of mastitis in cows" reference number AWEEC/BAU/2020(44) was approved by the Animal Welfare and Experimentation Ethics Committee, Bangladesh Agricultural University, Mymensingh-2202, Bangladesh.
Sample collection
Milk samples were obtained from 36-month and 46-month-old female lactating Holstein Friesian (Bos taurus taurus) cows as previously reported [13]. Cow udders were washed with clean water and dried, then the udder teats were rubbed with 70% ethanol. The first two strings were discarded, and California mastitis test was performed to determine the milk somatic cell counts in milk samples as previously reported [13]. Milk samples (10 mL) were collected in sterile tubes and samples were transported to the laboratory maintaining a cold chain for further analysis. Somatic cell counting was performed using Lactoscan Combo's SCC (Milkotronic Ltd, Bulgaria) according to the manufacturer's protocol.
Bacterial isolation
Milk samples (500 µL) were inoculated in 10 mL nutrient broth and incubated at 37°C for 18 hours and subsequently streaked on mannitol salt agar media (HiMedia). The inoculated plates were incubated at 37 °C for 24 hours and sub-cultured to isolate presumptively identified S. aureus pure colonies as previously reported [13].
DNA extraction and bacterial identification using PCR
Presumptive S. aureus pure single colonies were confirmed using S. aureus primer-specific PCR. Genomic DNA was extracted using genomic DNA Purification Kit (Promega, WI, USA) and isolates were confirmed by PCR using species-specific primers (GCG ATT GAT GGT GAT ACG GTT and AGC CAA GCC TTG ACG AAC TAA AGC) targeting the nuc gene as previously reported [14].
Antimicrobial Sensitivity testing
Antimicrobial susceptibility profiles against ciprofloxacin (5µg), cefoxitin (30µg), chloramphenicol (10µg), doxycycline (30µg), fosfomycin (50µg), gentamicin (10µg), levofloxacin (5µg), sulfamethoxazole - trimethoprim (1.25/23.75µg), and tetracycline (30µg) were determined using the Kirby-Bauer disk diffusion method (Oxoid Ltd., UK), as previously reported. The results were interpreted according to Clinical and Laboratory Standard Institute guidelines [15].
Whole-genome sequencing analysis
S. aureus PCR-confirmed isolates were submitted to Invent Technology Ltd. (Banani, Dhaka, Bangladesh) for whole-genome sequencing as recently reported [13]. Sequencing libraries were prepared using a Nextera XT library preparation kit (Illumina Inc., CA, USA) and sequencing was performed on the Illumina NextSeq 550 platform (Illumina Inc., CA, USA) using the high-output reagent kit with 150 nt maximal read length.
Bioinformatic analyses
Gene predictions and annotations were performed using the National Center for Biotechnology Information (NCBI) Prokaryotic Genome Annotation Pipeline (PGAP) [16]. Raw paired-end reads were quality checked and assembled to contigs using the ASA3P pipeline [17]. The detection of antibiotic resistance genes was performed using Resfinder 4.0 [18]. SCCmec type determination was performed using the SCCmecFinder tool, (https://cge.food.dtu.dk/services/SCCmecFinder/). Multilocus sequence type determination was performed using PubMLST [19]. spa type determination was performed using spaTyper 1.0 [20]. Virulence gene determination was performed using VFanalyzer [21]. The presence of Cap5A-P proteins from S. aureus USA300_FPR3757 [22] was determined using tblastN. Core-genome-based analysis was performed using ParSNP of the Harvest Suite package [23]. The resulting trees were annotated using ITol v 6.6 [24].
Results and Discussion
In the present study, a total of 423 randomly selected lactating cows were tested for the detection of S. aureus in their milk samples. It was found that 44.68% (189/423) of the cows were mastitis positive, of which 17.49% (74/423) were clinical and 27.19% (115/423) sub-clinical mastitis. Isolation of S. aureus was performed using mannitol salt agar which were subsequently confirmed using nuc-gene specific PCR. It was found that 54.49% (103 out of 189 mastitis affected cows) were infected with Staphylococcus aureus.
Genome sequences of two S. aureus BR-MHR220 and BR-MHR281 strains were generated and raw paired-end reads (average read count 18,863,133; average coverage 1024×, average read length 135 nt) were quality checked and assembled to contigs using the ASA3P pipeline [17]. Assembly was performed using SPAdes v3.13.0 [25] integrated in ASA3P. Contigs smaller than 200 bp were discarded. Contigs were uploaded to NCBI and annotated using the NCBI Prokaryotic Genome Annotation Pipeline v6.0 [16].
For BR-MHR220 genome, a total number of 54 contigs and 2,815,914 bp, with a G+C content of 32.75%, and a N50 value of 321,760 bp was achieved. For BR-MHR281 genome, a total number of 28 contigs and 2,728,146 bp, with a G+C content of 32.73%, and a N50 value of 470,470 bp was achieved.
Gene predictions and annotations identified 2,788 coding DNA sequences (CDS), 54 tRNAs, 4 ncRNA and 4 rRNA genes for BR-MHR220, and 2,677 coding DNA sequences, 56 tRNAs, 4 ncRNA and 4 rRNA genes for BR-MHR281.
Phenotypic antimicrobial profiling revealed that both BR-MHR220 and BR-MHR281 strains were resistant to cefoxitin, methicillin, and oxacillin. Additionally, BR-MHR220 strain was resistant to linezolid and penicillin G, while BR-MHR281 strain was resistant to gentamicin and levofloxacin. Both BR-MHR220 and BR-MHR281 strains were sensitive to chloramphenicol, ciprofloxacin, doxycycline, and sulfamethoxazole-trimethoprim. Additionally, BR-MHR220 strain was sensitive to gentamicin and levofloxacin, while BR-MHR281 strain was sensitive to linezolid, penicillin G, and tetracycline.
Genome analyses of the two BR-MHR220 and BR-MHR281 strains were performed and no antibiotic resistance genes were detected in strain BR-MHR281, however strain BR-MHR220 harbored the mecA and blaZ genes. The mecA gene is presumably located on a SCCmec type IVa(2B) element. The reported genotype did not correlate with the phenotypic resistance detected. The discrepancy between genotypic and phenotypic resistance may be explained by possible mutations of porin genes or overexpression of efflux pumps.
Only few S. aureus STs were previously reported to be positive for blaZ or mecA. Some human-adapted lineages, such as ST-5 (CC5), ST-8 (CC8), and their variants were previously isolated from cows and were positive for blaZ and mecA [26-28]. In the present study, we reported the detection of ST-6 (CC5) isolate positive for both blaZ and mecA.
Determination of virulence genes was performed using VFanalyzer [20]. In addition, the presence of Cap5A-P proteins from S. aureus USA300_FPR3757 [21] in both BR-MHR220 and BR-MHR281 genomes was determined using tblastN. The overview of detected virulence genes is depicted in Supplementary Table 1. Homologues of 60 virulence genes were detected in both isolates, while specific 19 and 13 virulence genes were only detected in BR-MHR220 and BR-MHR281, respectively. The virulence genes detected only in BR-MHR220 were the cell wall associated fibronectin binding protein ebh, the collagen adhesion cna, the intracellular adhesin icaD, two Ser-Asp rich fibrinogen-binding proteins (sdrD, sdrE), six serine proteases (splA, splB, splC, splD, splE, splF), Staphylokinase (sak), SCIN (scn), Enterotoxin A (sea), four Exotoxins (set7, set15, set16, set25) and Leukotoxin D (lukD). BR-MHR281 harbored seven enterotoxins (seg, yent2, selk, selm, seln, selo, selq). All were different from the one found in BR-MHR220. Five exotoxins were detected only in BR-MHR281 (set17, set21, set26, set30, set39). The toxic shock syndrome toxin (tsst) was detected only in BR-MHR281.
Multilocus sequence type determination was performed using PubMLST [18]. spa type determination was performed using spaTyper 1.0 [19]. BR-MHR220 was assigned to sequence type (ST-6), clonal complex (CC) 5 and spa type t304, while BR-MHR281 belonged to ST-2454 and CC8 and harbored the spa type t7867.
Several bovine-adapted S. aureus lineages including CC97, CC133, and CC151 and human-adapted lineages including CC1, CC5, CC8, CC30, and CC45 were previously reported [29]. In the present study, two strains belonging to CC5 and CC8 S. aureus lineages were isolated from bovine mastitis samples. Similarly, the genome sequences of three human-adapted isolates (two from CC97 and one from CC8), isolated from bovine mastitis samples were previously reported [30]. Altogether, the results provide significant insights on the role of genomic characteristics in early S. aureus host spillover events and the subsequent adaption to a new host. The zooanthroponotic transfer and the spillover transmission of CC5 and CC8 from humans to bovine in the present study are possible events that require further investigation and comparative genomic analysis.
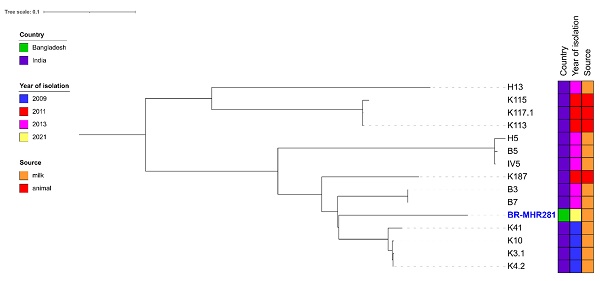
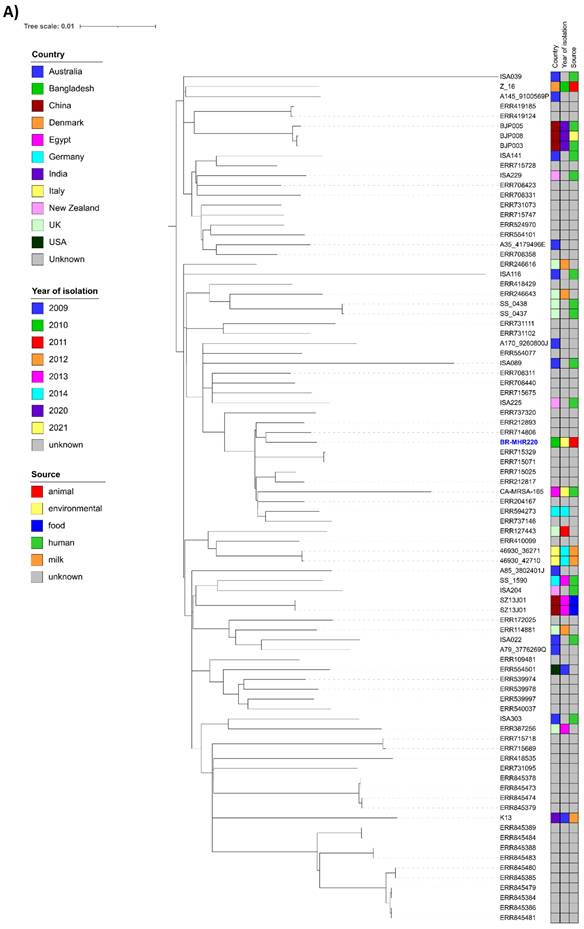
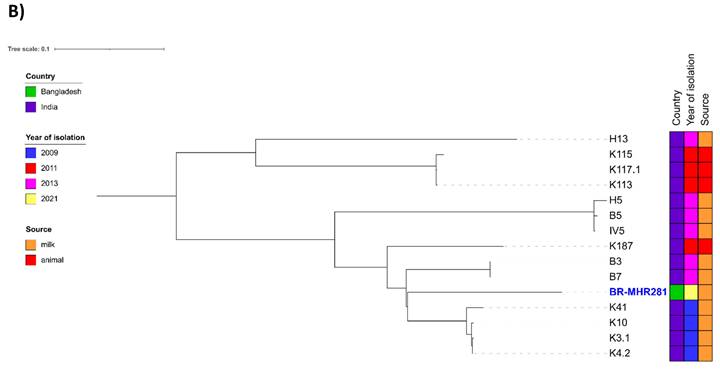
For comparison of BR-MHR220 and BR-MHR281 with global isolates, ST-6 (n=85, Supplementary Table 2) and ST-2454 (n= 14, Supplementary Table 3) isolate contigs were downloaded from the PubMLST database (as of 23rd January 2023). Core-genome-based analysis was performed using ParSNP of the Harvest Suite package [22]. The resulting trees were annotated using ITol v 6.6 [23]. As depicted in Figure 1A, the closest relative to BR-MHR220 was ERR714806, an isolate collected in the frame of a study on MRSA in England (source unknown) [31]. The closest relative to BR-MHR281 (Figure 1B) was K4.2, isolated from cow milk in India in 2009. The current study describes the use of whole genome sequencing (WGS) methods in the detection of bovine mastitis-associated S. aureus isolates in dairy cows in Bangladesh. The findings corroborate the worldwide distribution of S. aureus CC8 and CC5 isolates in different host species. At present however, WGS still remains unaffordable and inaccessible tool in resource-limited settings. The genome sequences of S. aureus strains in the present study will contribute to advanced understanding of the virulence, host adaptation, zoonotic and zooanthroponotic potential of S. aureus.
Phylogenetic trees of S. aureus strain (A) BR-MHR220 and (B) BR-MHR281. For the core genome-based comparison, ST-6 or ST-2454 sequence information present in PubMLST was used. Trees were annotated using ITOL v. 6.6 and modified using Inkscape.
Data availability
This whole-genome sequencing project has been deposited at DDBJ/ENA/GenBank under the BioProject number PRJNA716986 (BioSample accession numbers SAMN26025965 and SAMN26025969 and GenBank accession numbers JALBGM000000000 and JALBGI000000000. The versions described in this paper are the first version. The sequences have been submitted to the Sequence Read Archive (SRA) under the accession numbers SRR18182112 and SRR18182108. All isolates used in this study were submitted to Public Databases for molecular typing and microbial genome diversity for curation (https://pubmlst.org/organisms/staphylococcus-aureus) and are publicly available under PubMLST ID numbers 38059 and 38060.
Supplementary Material
Supplementary tables.
Acknowledgements
The whole-genome sequencing work was supported in part by the Bangladesh Academy of Sciences and the United States Department of Agriculture (Project ID: BAS-USDA LS-26/2020) and the Bangladesh Agricultural University (BAU). Authors thank the NCBI GenBank submission staff for help during the genome upload, decontamination and deposition process.
Funding
This project was supported in part by the Bangladesh Academy of Sciences and the United States Department of Agriculture (Project ID: BAS-USDA LS-26/2020), the Bangladesh Agricultural University (BAU), the Hessian Ministry of Higher Education, Research and Arts within the project HuKKH (Hessisches Universitaeres Kompetenzzentrum Krankenhaus Hygiene), Germany.
Declaration
No other individual has any role in the study design, analysis of data, writing of the manuscript or decision to publish. The views, opinions, and/or findings expressed are those of the authors and should not be interpreted as representing the official views or policies of the U.S. Department of Agriculture or the U.S. Government.
Author contributions
M.H.R.: Conceptualization, Methodology, Investigation, Formal analysis. MEZ: Conceptualization, Methodology, Data curation, Formal analysis, Investigation, Visualization, Validation, Writing - original draft, Writing - review & editing, Project administration, Supervision; LF: Formal analysis, Methodology, Visualization, Data curation, Software, Validation; Writing - original draft, Writing - review & editing; M.F.R.K.: Methodology, Investigation; J.A.: Methodology, Investigation; N.N.P.: Methodology, Investigation. H.M.A.: Conceptualization, Data curation, Formal analysis, Investigation, Visualization, Validation, Writing -original draft, Writing - review & editing; M.B.R.: Conceptualization, Formal analysis, Validation, Funding acquisition, Supervision, Project administration.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Halasa T, Huijps K, Østerås O, Hogeveen H. Economic effects of bovine mastitis and mastitis management: A review. Vet. Q. 2007;29:18-31
2. Pascu C, Herman V, Iancu I, Costina L. Etiology of Mastitis and Antimicrobial Resistance in Dairy Cattle Farms in the Western Part of Romania. Antibiotics. 2022;11:57
3. Fetsch A, Johler S. Staphylococcus aureus as a foodborne pathogen. Curr Clin Microbiol. 2018;5(2):88-96
4. Cremonesi P, Pozzi F, Raschetti M, Bignoli G, Capra E, Graber HU, Vezzoli F, Piccinini R, Bertasi B, Biffani S, Castiglioni B, Luini M. Genomic characteristics of Staphylococcus aureus strains associated with high within-herd prevalence of intramammary infections in dairy cows. J Dairy Sci. 2015;98(10):6828-38 doi: 10.3168/jds.2014-9074
5. Annamanedi M, Sheela P, Sundareshan S. et al. Molecular fingerprinting of bovine mastitis-associated Staphylococcus aureus isolates from India. Sci Rep. 2021;11:15228 doi: 10.1038/s41598-021-94760-x
6. Watts JL. Etiological agents of bovine mastitis. Vet Microbiol. 1988;16:41-66 doi: 10.1016/0378-1135(88)90126-5
7. Chan CX, Beiko RG, Ragan MA. Lateral transfer of genes and gene fragments in Staphylococcus extends beyond mobile elements. J. Bacteriol. 2011;193:3964-3977
8. Castelani L. et al. Molecular typing of mastitis causing Staphylococcus aureus isolated from heifers and cows. Int J Mol Sci. 2013;14:4326-4333
9. Neelam Jain VK, Singh M Joshi VG, Chhabra R Singh K. et al. Virulence and antimicrobial resistance gene profiles of Staphylococcus aureus associated with clinical mastitis in cattle. PLoS ONE. 2022;17(5):e0264762 doi: 0.1371/journal.pone.0264762
10. Maity S, Ambatipudi K. Mammary microbial dysbiosis leads to the zoonosis of bovine mastitis: a One-Health perspective. FEMS Microbiology Ecology. 2021;97(1):fiaa241 doi: 10.1093/femsec/fiaa241
11. de-Jong A, Garch FE, Simjee S, Moyaert H, Rose M, Youala M. et al. Monitoring of antimicrobial susceptibility of udder pathogens recovered from cases of clinical mastitis in dairy cows across Europe: Vet Path results. Vet Microbiol. 2018;213:73-81 pmid:29292007
12. Hoque MN, Istiaq A, Clement RA, Sultana M, Crandall KA, Siddiki AZ, Hossain MA. 2019. Metagenomic Deep Sequencing Reveals Association of Microbiome Signature with Functional Biases in Bovine Mastitis. Scientific Reports 9:13536.
13. Rahman MH, El Zowalaty ME, Falgenhauer L, Khan MFR, Alam J, Popy NN, Rahman MB. Draft Genome Sequences of Two Clinical Mastitis-Associated Escherichia coli Strains, of Sequence Type 101 and Novel Sequence Type 13054, Isolated from Dairy Cows in Bangladesh. Microbiol Resour Announc. 2023;12(8):e0016623 doi: 10.1128/mra.00166-23
14. Ewida RM, Al-Hosary AAT. Prevalence of enterotoxins and other virulence genes of Staphylococcus aureus caused subclinical mastitis in dairy cows. Veterinary World. 2020;13(6):1193-1198
15. CLSI (2020) CLSI M100-ED29. 2021 Performance Standards for Antimicrobial Susceptibility Testing, 30th Edition. Vol. 40, PA, USA.
16. Tatusova T, DiCuccio M, Badretdin A, Chetvernin V, Nawrocki EP, Zaslavsky L, Lomsadze A, Kim D, Borodovsky M, Ostell J. NCBI prokaryotic genome annotation pipeline. Nucleic Acids Res. 2016;44:6614-6624 doi: 10.1093/nar/gkw569
17. Schwengers O, Hoek A, Fritzenwanker M, Falgenhauer L, Hain T, Chakraborty T, Goesmann A. ASA3P: An automatic and scalable pipeline for the assembly, annotation and higher-level analysis of closely related bacterial isolates. PLoS Comput Biol. 2020;16(3):e1007134 doi: 10.1371/journal.pcbi.1007134
18. Bortolaia V, Kaas RS, Ruppe E, Roberts MC, Schwarz S, Cattoir V, Philippon A, Allesoe RL, Rebelo AR, Florensa AF, Fagelhauer L, Chakraborty T, Neumann B, Werner G, Bender JK, Stingl K, Nguyen M, Coppens J, Xavier BB, Malhotra-Kumar S, Westh H, Pinholt M, Anjum MF, Duggett NA, Kempf I, Nykäsenoja S, Olkkola S, Wieczorek K, Amaro A, Clemente L, Mossong J, Losch S, Ragimbeau C, Lund O, Aarestrup FM. ResFinder 4.0 for predictions of phenotypes from genotypes. J Antimicrob Chemother. 2020;75(12):3491-3500 doi: 10.1093/jac/dkaa345
19. Enright MC, Day NP, Davies CE, Peacock SJ, Spratt BG. Multilocus sequence typing for characterization of methicillin-resistant and methicillin-susceptible clones of Staphylococcus aureus. J Clin Microbiol. 2000;38(3):1008-15 doi: 10.1128/JCM.38.3.1008-1015.2000
20. Bartels MD, Petersen A, Worning P, Nielsen JB, Larner-Svensson H, Johansen HK, Andersen LP, Jarløv JO, Boye K, Larsen AR, Westh H. Comparing whole-genome sequencing with Sanger sequencing for spa typing of methicillin-resistant Staphylococcus aureus. J Clin Microbiol. 2014;52(12):4305-8 doi: 10.1128/JCM.01979-14
21. Liu B, Zheng D, Jin Q, Chen L, Yang J. VFDB 2019: a comparative pathogenomic platform with an interactive web interface. Nucleic Acids Res. 2019;47(D1):D687-D692 doi: 10.1093/nar/gky1080
22. Diep BA, Gill SR, Chang RF, Phan TH, Chen JH, Davidson MG, Lin F, Lin J, Carleton HA, Mongodin EF, Sensabaugh GF, Perdreau-Remington F. Complete genome sequence of USA300, an epidemic clone of community-acquired meticillin-resistant Staphylococcus aureus. Lancet. 2006;367(9512):731-9 doi: 10.1016/S0140-6736(06)68231-7
23. Treangen TJ, Ondov BD, Koren S, Phillippy AM. The Harvest suite for rapid core-genome alignment and visualization of thousands of intraspecific microbial genomes. Genome Biol. 2014;15(11):524 doi: 10.1186/s13059-014-0524-x
24. Letunic I, Bork P. Interactive Tree Of Life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 2021;49(W1):W293-W296 doi: 10.1093/nar/gkab301
25. Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M, Kulikov AS, Lesin VM, Nikolenko SI, Pham S, Prjibelski AD, Pyshkin AV, Sirotkin AV, Vyahhi N, Tesler G, Alekseyev MA, Pevzner PA. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol. 2012;19(5):455-77 doi: 10.1089/cmb.2012.0021
26. Klibi A, Jouini A, Gómez P, Slimene K, Ceballos S. et al. Molecular characterization and clonal diversity of methicillin-resistant and -susceptible Staphylococcus aureus isolates of milk of cows with clinical mastitis in Tunisia. Microb Drug Resist. 2018;24:1210-1216 doi: 10.1089/mdr.2017.0278
27. Schmidt T, Kock MM, Ehlers MM. Molecular characterization of Staphylococcus aureus isolated from bovine mastitis and close human contacts in South African dairy herds: genetic diversity and inter-species host transmission. Front Microbiol. 2017;8:511 doi: 10.3389/fmicb.2017.00511
28. Käppeli N, Morach M, Corti S, Eicher C, Stephan R. et al. Staphylococcus aureus related to bovine mastitis in Switzerland: Clonal diversity, virulence gene profiles, and antimicrobial resistance of isolates collected throughout 2017. J Dairy Sci. 2019;102:3274-3281 doi: 10.3168/jds.2018-15317
29. Park S, Ronholm J. Staphylococcus aureus in agriculture: lessons in evolution from a multispecies pathogen. Clin Microbiol Rev. 2021;34:e00182-20 doi: 10.1128/CMR.00182-20
30. Park S, Jung D, O'Brien B, Ruffini J, Dussault F, Dube-Duquette A, Demontier É, Lucier JF, Malouin F, Dufour S, Ronholm J. Comparative genomic analysis of Staphylococcus aureus isolates associated with either bovine intramammary infections or human infections demonstrates the importance of restriction-modification systems in host adaptation. Microb Genom. 2022Feb;8(2):000779 doi: 10.1099/mgen.0.000779
31. Toleman MS, Reuter S, Coll F, Harrison EM, Blane B, Brown NM, Török ME, Parkhill J, Peacock SJ. Systematic Surveillance Detects Multiple Silent Introductions and Household Transmission of Methicillin-Resistant Staphylococcus aureus USA300 in the East of England. J Infect Dis. 2016Aug1;214(3):447-53 doi: 10.1093/infdis/jiw166
Author contact
Corresponding author: MEZ; elzow005com and bahanurredu.bd.

 Global reach, higher impact
Global reach, higher impact